Current worldwide clinical trials on COVID-19
dimensions.ai provides a daily updated list of clinical trials for COVID-19.
Analysis of Clinical Trial Data
Using dimensions.ai service and simple NLP scripts, the following figures show the distribution of COVID-19 clinical trials around the world and the different types of trials. These statistics are synchronized with each update from the figshare table.
Figures Last Updated: February 21, 2021
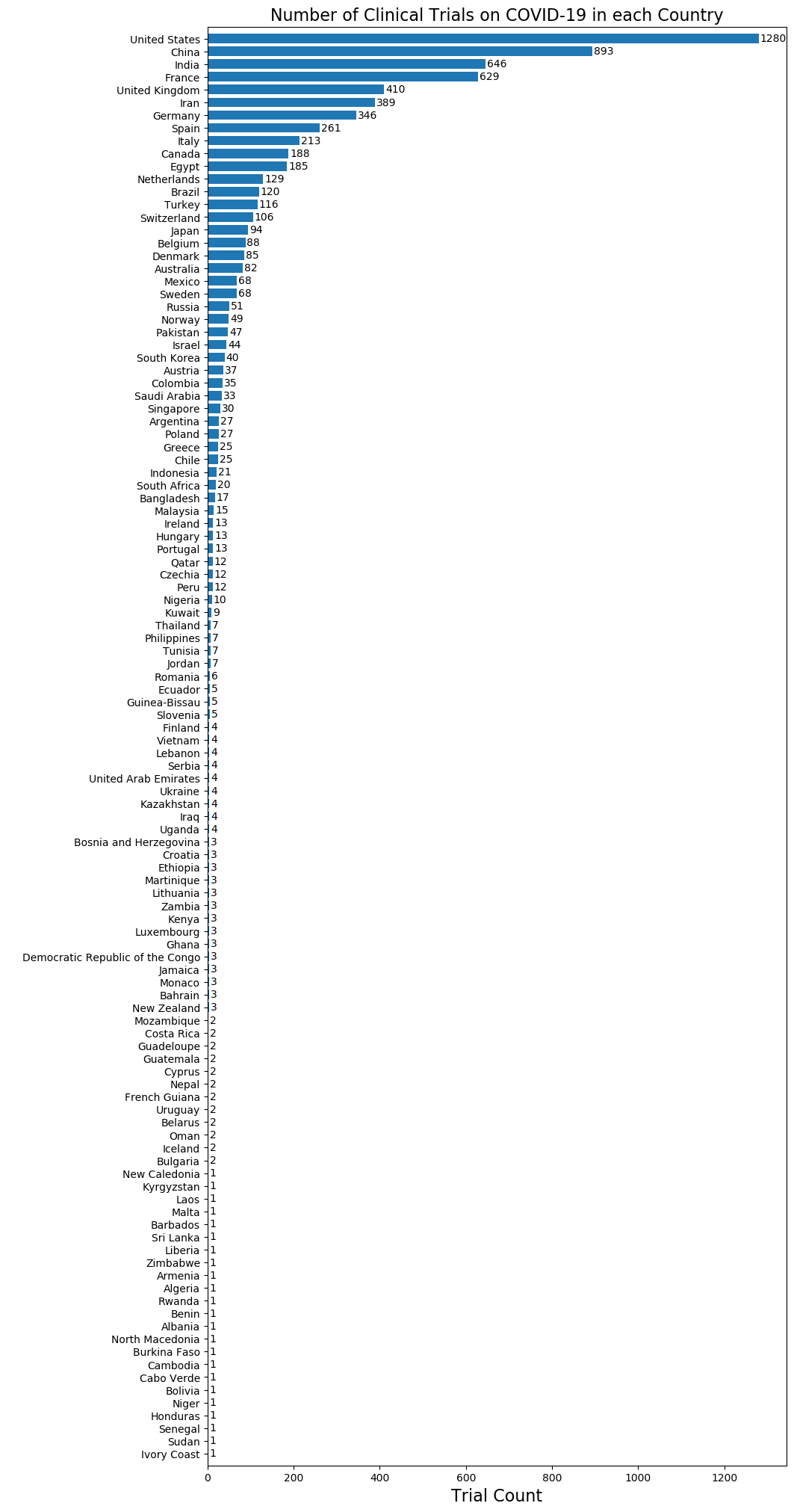
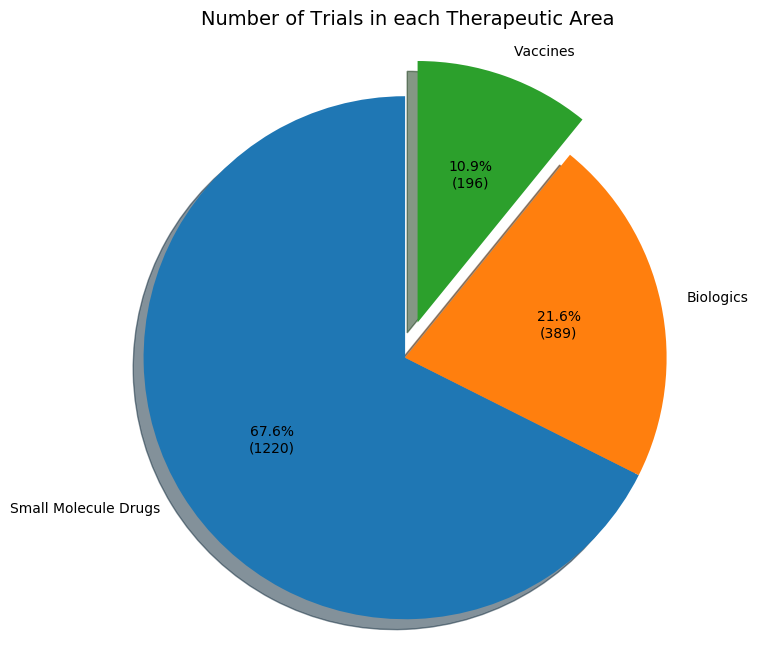
Download links for all three types of clinical trials are listed below.
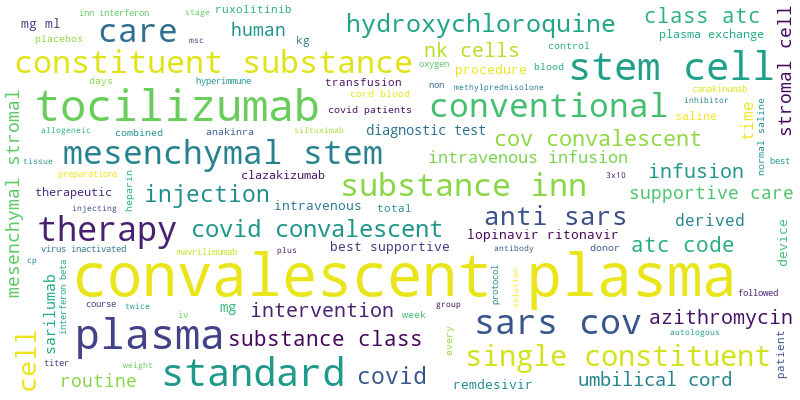
Download Biologics Clinical Trial list, Word Cloud below:
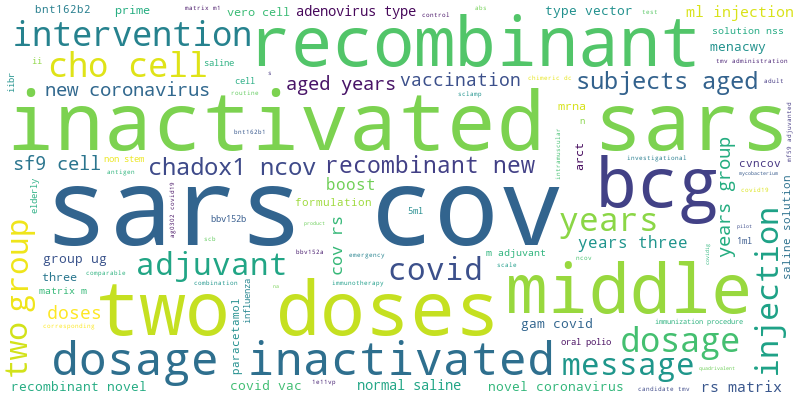
Download Vaccine Clinical Trial list, Word Cloud below:
Similarily, using the same data, a preliminary small molecule treatment list with clinical trial counts was automatically generated using relevant key words. This list is only an estimate and will be updated daily. The list for this figure is available for download in the Treatment list link above.
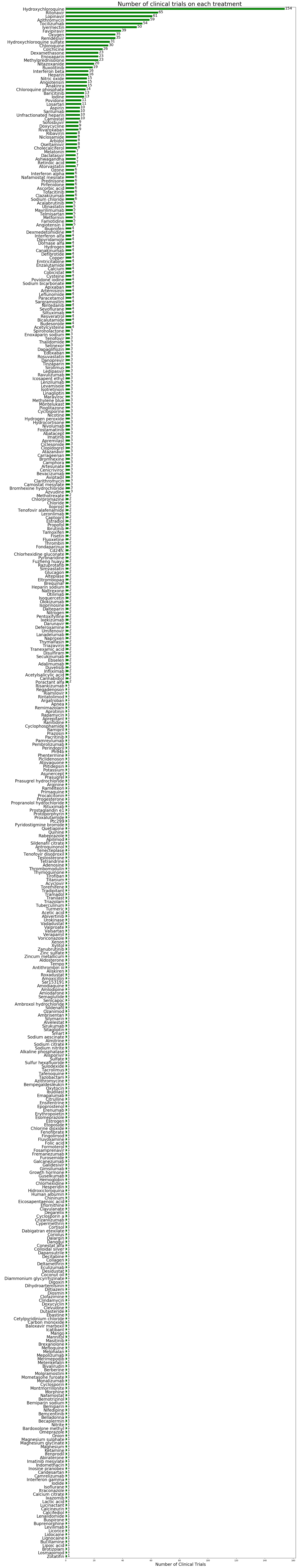
Last update: September 15, 2022
Your feedback is highly appreciated.